Dr. Xiaoyu Hu’s group reveals a critical role for creatine in regulating macrophage polarization
Source:Xiaoyu Hu
2019-08-22
On August 20, 2019, Dr. Xiaoyu Hu’s group at the Tsinghua University Institute for Immunology published a research article entitled “Slc6a8-mediated creatine uptake and accumulation reprogram macrophage polarization via regulating cytokine responses” in Immunity. This work revealed that creatine reprogrammed macrophage polarization by suppressing IFN-γ-induced macrophage activation yet promoting IL-4-activated macrophage polarization.
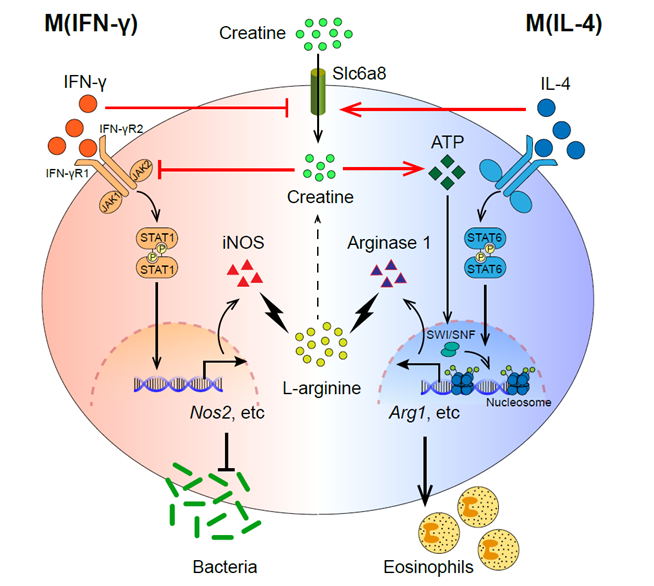
Macrophage polarization is accompanied by drastic changes of L-arginine metabolism. Two L-arginine catalytic enzymes, iNOS and arginase 1, are well-characterized hallmark molecules of classically and alternatively activated macrophages, respectively. The third metabolic fate of L-arginine is generation of creatine that acts as a key source of cellular energy reserve, yet little is known about the role of creatine in the immune system. Researchers from Hu’s and Chen’s groups combined genetic, genomic, metabolic and immunological analyses and revealed that creatine reprogrammed macrophage polarization by suppressing M(IFN-γ) yet promoting M(IL-4) effector functions. Mechanistically, creatine inhibited induction of immune effector molecules including iNOS by suppressing IFN-γ-JAK-STAT1 transcription factor signaling while supporting IL-4-STAT6-activated arginase 1 expression by promoting chromatin remodeling. Depletion of intracellular creatine by ablation of creatine transporter Slc6a8 altered macrophage-mediated immune responses in vivo. These results uncover a previously uncharacterized role for creatine in macrophage polarization by modulating cellular responses to cytokines such as IFN-γ and IL-4. These findings connected two seemingly irrelevant biological processes, namely creatine metabolism and macrophage polarization, and implicate creatine as a key player in the immunometabolism arena.
Dr. Xiaoyu Hu and Dr. Ligong Chen are the corresponding authors of this article. Ph.D. student Liangliang Ji and Dr. Xinbin Zhao are the co-first authors. Ph.D. students Bin Zhang, Lan Kang and Wenxin Song also contributed to this study. This study was supported by Ministry of Science and Technology of China (National Key Research Project), National Natural Science Foundation of China, Nation Science and Technology Major Projects for Major New Drugs Innovation and Development, Tsinghua University Initiative Scientific Research Program, funds from Tsinghua-Peking Center for Life Sciences, Institute for Immunology at Tsinghua University.
Links: https://www.sciencedirect.com/science/article/pii/S1074761319302778
Macrophage polarization is accompanied by drastic changes of L-arginine metabolism. Two L-arginine catalytic enzymes, iNOS and arginase 1, are well-characterized hallmark molecules of classically and alternatively activated macrophages, respectively. The third metabolic fate of L-arginine is generation of creatine that acts as a key source of cellular energy reserve, yet little is known about the role of creatine in the immune system. Researchers from Hu’s and Chen’s groups combined genetic, genomic, metabolic and immunological analyses and revealed that creatine reprogrammed macrophage polarization by suppressing M(IFN-γ) yet promoting M(IL-4) effector functions. Mechanistically, creatine inhibited induction of immune effector molecules including iNOS by suppressing IFN-γ-JAK-STAT1 transcription factor signaling while supporting IL-4-STAT6-activated arginase 1 expression by promoting chromatin remodeling. Depletion of intracellular creatine by ablation of creatine transporter Slc6a8 altered macrophage-mediated immune responses in vivo. These results uncover a previously uncharacterized role for creatine in macrophage polarization by modulating cellular responses to cytokines such as IFN-γ and IL-4. These findings connected two seemingly irrelevant biological processes, namely creatine metabolism and macrophage polarization, and implicate creatine as a key player in the immunometabolism arena.

Dr. Xiaoyu Hu and Dr. Ligong Chen are the corresponding authors of this article. Ph.D. student Liangliang Ji and Dr. Xinbin Zhao are the co-first authors. Ph.D. students Bin Zhang, Lan Kang and Wenxin Song also contributed to this study. This study was supported by Ministry of Science and Technology of China (National Key Research Project), National Natural Science Foundation of China, Nation Science and Technology Major Projects for Major New Drugs Innovation and Development, Tsinghua University Initiative Scientific Research Program, funds from Tsinghua-Peking Center for Life Sciences, Institute for Immunology at Tsinghua University.
Links: https://www.sciencedirect.com/science/article/pii/S1074761319302778


