Prof. Xuetao Cao’s group found a new mechanism for host cells to resist viral infection
Source:Yang Liu
2019-08-26
On August 23rd, Prof. Xuetao Cao’s group published an article in Science entitled “N6-methyladenosine RNA modification-mediated cellular metabolism rewiring inhibits viral replication”, reported that host cells can regulate m6A RNA modification and metabolism rewiring to resist viral infection.
This finding reveals a new mechanism by which host cells can resist viral infections through an innate immune-independent manner, helping us to understand the virus-host interaction and its molecular mechanisms, providing intervention and treatment for viral infectious diseases.
Host cells can inhibit viral infection via a variety of ways, for example, by producing interferon during innate immune response. However, whether host cells can resist viral infection through innate immune-independent manner and related mechanisms still remains unknown.
By performing a series of in vitro functional screening and in vivo experiments, authors found that RNA m6A demethylase ALKBH5 has a broad effect on promoting replication of different types of viruses. Furthermore, RNA-seq, m6A-seq, iCLIP-seq, metabolomics analysis and anti-viral function screening identified that ALKBH5 takes effect mainly by regulating the cellular metabolism rewiring.
This study elucidated that host cells actively respond to viral infection by impairing m6A demethylation activity of ALKBH5, leading to the increased mRNA decay and reduced protein expression of metabolic enzyme OGDH, consequently reprogramming cellular metabolic state to confer IFN-I-independent host resistance to viral infection.
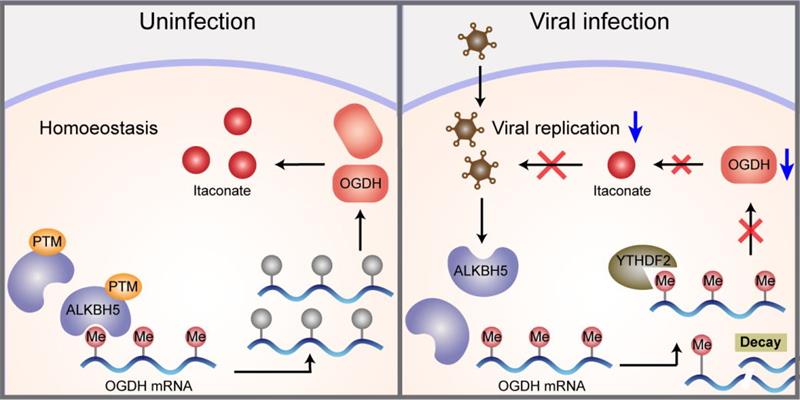
The findings on the crosstalk of m6A RNA modification and metabolic reprogramming via the ALKBH5-OGDH-Itaconate pathway provide mechanistic insight into the cellular defense against invading pathogens through the innate response-independent metabolism rewiring. Identifying the cellular metabolites in host-virus interaction contributes to better understanding of viral infection process and helps identify potential targets for intervention.
Links: http://science.sciencemag.org/lookup/doi/10.1126/science.aax4468
This finding reveals a new mechanism by which host cells can resist viral infections through an innate immune-independent manner, helping us to understand the virus-host interaction and its molecular mechanisms, providing intervention and treatment for viral infectious diseases.
Host cells can inhibit viral infection via a variety of ways, for example, by producing interferon during innate immune response. However, whether host cells can resist viral infection through innate immune-independent manner and related mechanisms still remains unknown.
By performing a series of in vitro functional screening and in vivo experiments, authors found that RNA m6A demethylase ALKBH5 has a broad effect on promoting replication of different types of viruses. Furthermore, RNA-seq, m6A-seq, iCLIP-seq, metabolomics analysis and anti-viral function screening identified that ALKBH5 takes effect mainly by regulating the cellular metabolism rewiring.
This study elucidated that host cells actively respond to viral infection by impairing m6A demethylation activity of ALKBH5, leading to the increased mRNA decay and reduced protein expression of metabolic enzyme OGDH, consequently reprogramming cellular metabolic state to confer IFN-I-independent host resistance to viral infection.

The findings on the crosstalk of m6A RNA modification and metabolic reprogramming via the ALKBH5-OGDH-Itaconate pathway provide mechanistic insight into the cellular defense against invading pathogens through the innate response-independent metabolism rewiring. Identifying the cellular metabolites in host-virus interaction contributes to better understanding of viral infection process and helps identify potential targets for intervention.
Links: http://science.sciencemag.org/lookup/doi/10.1126/science.aax4468


