Bo Huang’s group reports ketone body metabolism regulates the formation and maintenance of memory T cell
Source:Huafeng Zhang
2019-12-31
Prof. Bo Huang (Peking Union Medical College & Chinese Academy of Medical Sciences) and his group found that the ketone body metabolism of memory T cells is active. as an epigenetic regulator, β-hydroxybutyric acid, the main metabolite, plays an important role in the development, maintenance and long-term survival of memory T cells. The related research paper (Ketogenesis-genera9ted β-hydroxybutyrate is an epigenetic regulator of CD8+ T cell memory development) was published online in Nature Cell Biology on December 23rd.
Glycogen is a polymer of glucose, mainly found in hepatocytes and muscle cells, which is used to maintain the stability of glucose concentration in the body's blood. When glucose levels are low or lack of energy, glucose is replenished by cell breakdown of glycogen, while when energy is sufficient, glucose is used to synthesize glycogen and is stored. For a long time, glycogen is only regarded as a storage molecule of glucose and participates in energy metabolism. Previous research by Professor Huang Bo's team found that memory T cells regulate the formation and maintenance of memory through glycogen metabolism. Memory T cells highly express cytoplasmic phosphoenolpyruvate carboxykinase (PCK1), a key rate-limiting enzyme in the gluconeogenesis pathway, which catalyzes the production of oxaloacetic acid to glucose-6-phosphate, but the glucose-6-phosphate is not converted to glucose, but to the synthesis of glycogen. The synthesized glycogen is decomposed and regenerated into glucose 6-phosphate, while glucose-6-phosphate enters the pentose phosphate pathway, producing reduced NADPH, to maintain a high level of reduced glutathione, scavenging intracellular free radicals in time, thus maintaining the long-term survival of memory T cells. The results were published in the journal Nature Cell Biology in 2018.
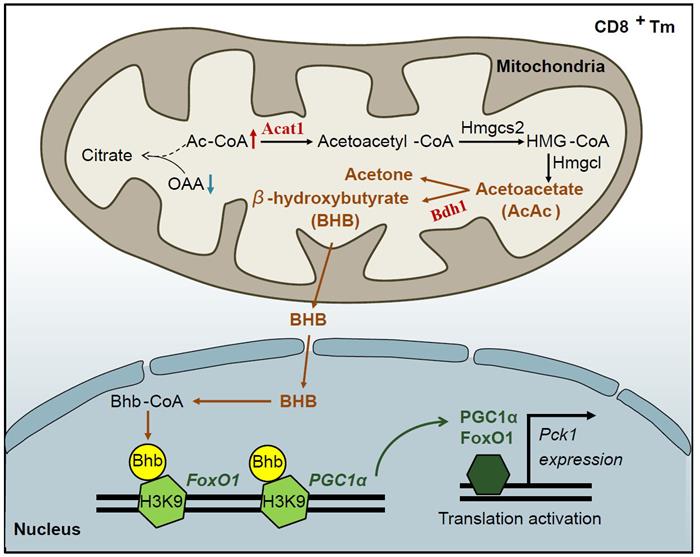
Despite the above elucidation of the metabolic pattern of memory T cells, the mechanism by which PCK1, a key enzyme that initiates the whole memory metabolism, is up-regulated in memory T cells is still unclear. In this study, the team found that acetyl-CoA (acetyl-CoA), a β-oxidized (Fatty acid beta-oxidation,FAO metabolite of fatty acids in the mitochondria of memory T cells, could enter the ketone body metabolism and up-regulate the expression of PCK1 through epigenetic modification.
Ketone bodies are the energy molecules of the body in a state of long-term hunger or disease, such as liver failure. It is generally believed that hepatocytes express enzymes related to the production of ketone bodies, but do not express enzymes for the cleavage of ketone bodies, so that the synthetic ketone bodies are effectively exported for use in extrahepatic tissues. Tricarboxylic acid cycle is the basic way of cell energy supply, and the polymerization of oxaloacetic acid and acetyl-CoA to citric acid is its initial step.
In this study, it was found that the large consumption of oxaloacetic acid to gluconeogenesis in CD8+ memory T cells led to the accumulation of acetyl-CoA in mitochondria and then to the production of ketone bodies, including the production of β-hydroxybutyric acid, the main component of oxaloacetic acid. Through high precision mass spectrometry and ChIP-seq sequencing, it was found that β-hydroxybutyric acid as an epigenetic factor could modify histone H3K9, make it β-hydroxybutyryl, and then up-regulate the expression of genes FoxO1 and PGC1 α. FoxO1 and PGC-1 α act as co-transcriptional factors to up-regulate the expression of PCK1, and then drive the pathways of gluconeogenesis and glycogen metabolism to maintain the long-term survival of memory T cells.
Memory is the most fundamental feature of the immune system. Understanding the formation mechanism of immune memory not only enables us to better understand the working principle of the immune system, but also has important guiding significance for the current immunotherapy and vaccine research and development. According to the unique metabolic mode of memory T cells, it is expected to find potential targets and develop new immunotherapy methods and vaccines by regulating ketone body metabolism, gluconeogenesis or glycogen metabolism. It is worth noting that the metabolic behavior of memory T cells breaks our traditional understanding of glycogen metabolism: glycogen synthesis and decomposition are considered to be two antagonistic processes that can not occur at the same time. However, the study of glycogen metabolism in memory T cells shows that glycogen synthesis and decomposition can be carried out at the same time in specific cells.
The research is supported by the Medical and Health Science and Technology Innovation Engineering Foundation of the Chinese Academy of Medical Sciences and the National Natural Science Foundation of China.
Links: https://www.nature.com/articles/s41556-019-0440-0
Glycogen is a polymer of glucose, mainly found in hepatocytes and muscle cells, which is used to maintain the stability of glucose concentration in the body's blood. When glucose levels are low or lack of energy, glucose is replenished by cell breakdown of glycogen, while when energy is sufficient, glucose is used to synthesize glycogen and is stored. For a long time, glycogen is only regarded as a storage molecule of glucose and participates in energy metabolism. Previous research by Professor Huang Bo's team found that memory T cells regulate the formation and maintenance of memory through glycogen metabolism. Memory T cells highly express cytoplasmic phosphoenolpyruvate carboxykinase (PCK1), a key rate-limiting enzyme in the gluconeogenesis pathway, which catalyzes the production of oxaloacetic acid to glucose-6-phosphate, but the glucose-6-phosphate is not converted to glucose, but to the synthesis of glycogen. The synthesized glycogen is decomposed and regenerated into glucose 6-phosphate, while glucose-6-phosphate enters the pentose phosphate pathway, producing reduced NADPH, to maintain a high level of reduced glutathione, scavenging intracellular free radicals in time, thus maintaining the long-term survival of memory T cells. The results were published in the journal Nature Cell Biology in 2018.
Despite the above elucidation of the metabolic pattern of memory T cells, the mechanism by which PCK1, a key enzyme that initiates the whole memory metabolism, is up-regulated in memory T cells is still unclear. In this study, the team found that acetyl-CoA (acetyl-CoA), a β-oxidized (Fatty acid beta-oxidation,FAO metabolite of fatty acids in the mitochondria of memory T cells, could enter the ketone body metabolism and up-regulate the expression of PCK1 through epigenetic modification.
Ketone bodies are the energy molecules of the body in a state of long-term hunger or disease, such as liver failure. It is generally believed that hepatocytes express enzymes related to the production of ketone bodies, but do not express enzymes for the cleavage of ketone bodies, so that the synthetic ketone bodies are effectively exported for use in extrahepatic tissues. Tricarboxylic acid cycle is the basic way of cell energy supply, and the polymerization of oxaloacetic acid and acetyl-CoA to citric acid is its initial step.
In this study, it was found that the large consumption of oxaloacetic acid to gluconeogenesis in CD8+ memory T cells led to the accumulation of acetyl-CoA in mitochondria and then to the production of ketone bodies, including the production of β-hydroxybutyric acid, the main component of oxaloacetic acid. Through high precision mass spectrometry and ChIP-seq sequencing, it was found that β-hydroxybutyric acid as an epigenetic factor could modify histone H3K9, make it β-hydroxybutyryl, and then up-regulate the expression of genes FoxO1 and PGC1 α. FoxO1 and PGC-1 α act as co-transcriptional factors to up-regulate the expression of PCK1, and then drive the pathways of gluconeogenesis and glycogen metabolism to maintain the long-term survival of memory T cells.
Memory is the most fundamental feature of the immune system. Understanding the formation mechanism of immune memory not only enables us to better understand the working principle of the immune system, but also has important guiding significance for the current immunotherapy and vaccine research and development. According to the unique metabolic mode of memory T cells, it is expected to find potential targets and develop new immunotherapy methods and vaccines by regulating ketone body metabolism, gluconeogenesis or glycogen metabolism. It is worth noting that the metabolic behavior of memory T cells breaks our traditional understanding of glycogen metabolism: glycogen synthesis and decomposition are considered to be two antagonistic processes that can not occur at the same time. However, the study of glycogen metabolism in memory T cells shows that glycogen synthesis and decomposition can be carried out at the same time in specific cells.
The research is supported by the Medical and Health Science and Technology Innovation Engineering Foundation of the Chinese Academy of Medical Sciences and the National Natural Science Foundation of China.

Links: https://www.nature.com/articles/s41556-019-0440-0


