Prof. Zhigang Tian’s group reveals extramedullary development of innate lymphoid cells
Source:Hui Peng
2021-03-30
The predominant sites where hematopoiesis occurs change during ontogeny. It has been widely accepted that the bone marrow (BM) is the primary site of hematopoiesis during postnatal life, and development of immune cells depends on BM hematopoiesis. In 2013, Prof. Zhigang Tian’s group reported the existence of liver-resident NK cells, which do not circulate in the blood and differ from cNK cells in many aspects. These cells are also referred to as liver type 1 innate lymphoid cells (ILC1s). Although remarkable progress has been made in function and transcriptional regulation of ILC1s, their developmental origin remains unclear.
Recently, a research article entitled “Liver type 1 innate lymphoid cells develop locally via an interferon-γ-dependent loop” has been published in Science. The study shows that adult mouse liver contains progenitors of liver-resident ILC1s, revealing extramedullary development pathway of ILCs.
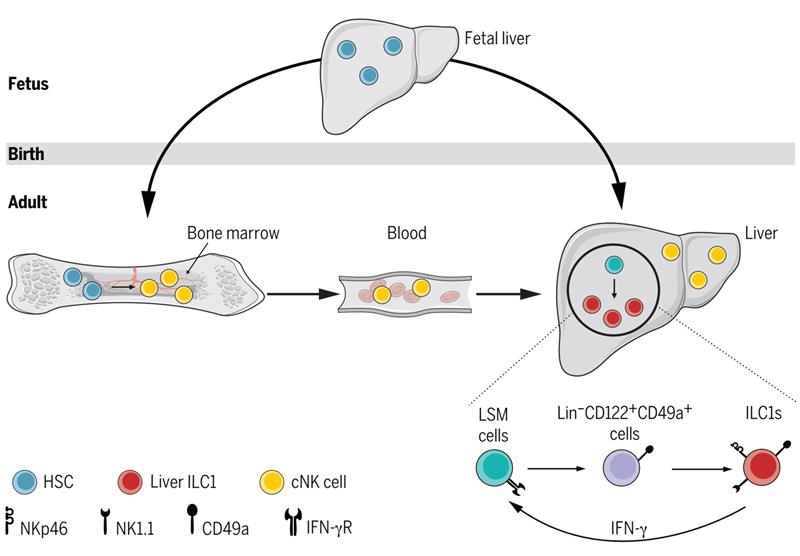
Given the existing knowledge about tissue-resident status of CD49a+CD49b− ILC1s in the liver, their impaired reconstitution in mice receiving BM transplants, and the adult liver environment compatible with hematopoiesis, the researchers asked whether liver ILC1s could develop from local hematopoietic progenitors during adulthood. By comparing the phenotype of lineage-negative hematopoietic progenitors in the adult liver, BM, and fetal liver, they found the adult liver contained Lin−Sca-1+Mac-1+ (LSM) cells resembling fetal liver hematopoietic stem cells. Adoptive transfer experiments revealed that LSM cells were able to generate multiple hematopoietic lineages, including Lin−CD122+CD49a+ progenitors that could further develop into liver ILC1s but not conventional NK cells. Mechanistically, the researchers found that deficiency in the gene encoding interferon-γ (IFN-γ) or one of its receptors selectively reduced the frequency and number of ILC1s and not cNK cells in the liver. IFN-γ production by the liver ILC1s themselves promoted the expansion and differentiation of LSM cells. Thus, these findings unveil an IFN-γ–dependent loop drives liver ILC1 development in situ, highlighting the contribution of extramedullary hematopoiesis to regional immune composition within the liver.
Prof. Zhigang Tian, Prof. Hui Peng, and Prof. Rui Sun from University of Science and Technology of China, and Prof. Eric Viver from Aix Marseille University are co-corresponding authors. Lu Bai and Ling Tang from University of Science and Technology of China, Margaux Vienne and Yann Kerdiles from Aix Marseille University are co-first authors. Funding sources of this research include the National Natural Science Foundation of China, National Key R&D Program of China, Chinese Academy of Sciences, CAMS Innovation Fund for Medical Sciences, etc.
Links: https://science.sciencemag.org/content/371/6536/eaba4177.full
Recently, a research article entitled “Liver type 1 innate lymphoid cells develop locally via an interferon-γ-dependent loop” has been published in Science. The study shows that adult mouse liver contains progenitors of liver-resident ILC1s, revealing extramedullary development pathway of ILCs.
Given the existing knowledge about tissue-resident status of CD49a+CD49b− ILC1s in the liver, their impaired reconstitution in mice receiving BM transplants, and the adult liver environment compatible with hematopoiesis, the researchers asked whether liver ILC1s could develop from local hematopoietic progenitors during adulthood. By comparing the phenotype of lineage-negative hematopoietic progenitors in the adult liver, BM, and fetal liver, they found the adult liver contained Lin−Sca-1+Mac-1+ (LSM) cells resembling fetal liver hematopoietic stem cells. Adoptive transfer experiments revealed that LSM cells were able to generate multiple hematopoietic lineages, including Lin−CD122+CD49a+ progenitors that could further develop into liver ILC1s but not conventional NK cells. Mechanistically, the researchers found that deficiency in the gene encoding interferon-γ (IFN-γ) or one of its receptors selectively reduced the frequency and number of ILC1s and not cNK cells in the liver. IFN-γ production by the liver ILC1s themselves promoted the expansion and differentiation of LSM cells. Thus, these findings unveil an IFN-γ–dependent loop drives liver ILC1 development in situ, highlighting the contribution of extramedullary hematopoiesis to regional immune composition within the liver.

Liver ILC1s develop in situ during adulthood
Prof. Zhigang Tian, Prof. Hui Peng, and Prof. Rui Sun from University of Science and Technology of China, and Prof. Eric Viver from Aix Marseille University are co-corresponding authors. Lu Bai and Ling Tang from University of Science and Technology of China, Margaux Vienne and Yann Kerdiles from Aix Marseille University are co-first authors. Funding sources of this research include the National Natural Science Foundation of China, National Key R&D Program of China, Chinese Academy of Sciences, CAMS Innovation Fund for Medical Sciences, etc.
Links: https://science.sciencemag.org/content/371/6536/eaba4177.full


