Nature Immunology Reported Novel Mechanisms and a Promising Target of Chronic Rhinosinusitis
Source:Yi Xu
2022-10-04
On Sep 22nd, 2022, Nature Immunology published an article entitled “Single-cell profiling identifies mechanisms of inflammatory heterogeneity in chronic rhinosinusitis”, which was a collaboration by Drs. Wei He & Jianmin Zhang group at the Institute of Basic Medical Science, Chinses Academy of Medical Science and Dr. Wei Lv group at Peking Union Medical College Hospital.
In this study, they performed a single-cell level analysis of immune and nonimmune cells in healthy individuals and patients with three subtypes of chronic rhinosinusitis (CRS) and identified some disease-specific cell subsets and molecules that specifically contribute to the pathogenesis. Notably, ALOX15+ macrophages were identified to make a major contribution to the pathogenesis of type 2 immunity in eosinophilic CRS with nasal polyps (eCRSwNP) by secreting chemokines that recruited eosinophils, monocytes and TH2 cells. The findings of this study not only provide a further understanding of the heterogeneous immune microenvironment and the pathogenesis of CRS subtypes but also identified novel targets for the development of therapeutic approaches for the treatment of CRS and potentially other type 2 immunity-mediated diseases.
Human airway chronic inflammatory diseases, including CRS and asthma, share specific mechanisms of pathogenesis and often coexist. Heterogeneous CRS can be divided into different subtypes, including CRS with nasal polyps (CRSwNP) and CRS without nasal polyps (CRSsNP), with the former further divided into eosinophilic CRSwNP (eCRSwNP) and non-eosinophilic CRSwNP (neCRSwNP). Current clinical guidelines still recommend local glucocorticosteroids as primary medication in the management of CRS and asthma, irrespective of disease subtypes. However, because of the heterogeneity of CRS and asthma subtypes with distinct immune responses and inflammatory profiles, such as type 2 and non-type 2 inflammation, the efficacy of these treatments is quite variable. The inflammatory microenvironment in CRS is complex and heterogeneous. Therefore, it would be meaningful to elevate treatment efficacy by modulating clinical therapeutic plan precisely according to systematic understandings for the compositions of immune microenvironment and pathogenesis mechanisms in different CRS subtypes. (Fig1)
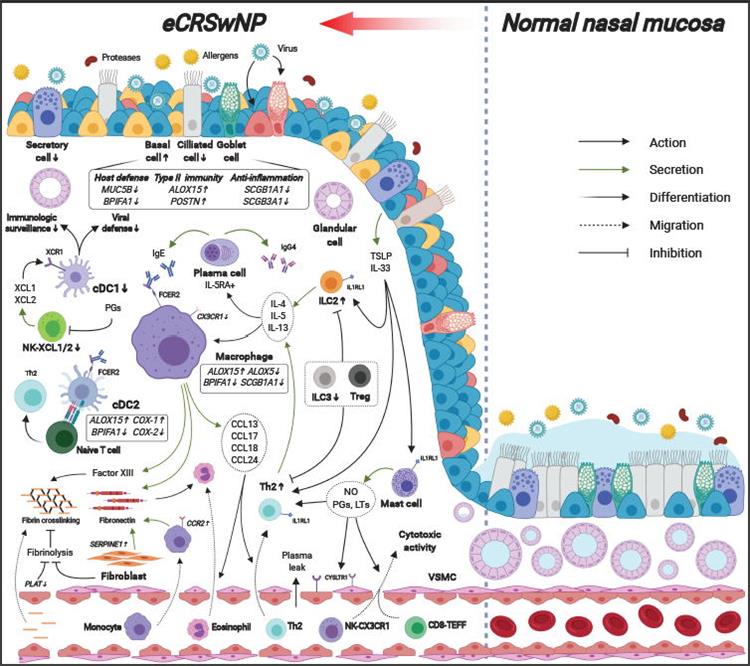
This work provided a detailed atlas of the immune and nonimmune cell microenvironment in the nasal mucosa of healthy individuals and patients with three heterogeneous subtypes of CRS at single-cell resolution, identifying novel disease-specific cell subsets and clusters, and key molecules associated with distinctive type 2 immunity related to eCRSwNP. The graphic summary illustrates novel insights on different nasal mucosa immune microenvironments between eCRSwNP and healthy controls, and how the type 2 immune response promotes the pathogenesis of nasal polyps in their scRNA-seq study. In eCRSwNP, the hallmarks of pathogenesis included: (1) hyperplasia of basal cells and deficiency of protective cells and molecules, (2) extracellular matrix remodeling dysfunction by fibroblasts, macrophages, vascular endothelial cells, and smooth muscle cells (SMCs), (3) elevated levels of TH2 cells, ILC2s, IL5RA+ plasma cells, and cytotoxic CX3CR1+CD8+ TEFF cells and NK cells, and deficiency of CD8+ TRM cells, (4) defects in the NK-cDC1 immunologic surveillance axis and overactivation of the ALOX15+ cDC2-TH2 axis, and very importantly, (5) enrichment of ALOX15+ macrophages with a prominent chemotactic effect as a driver of the type 2 immunity by promoting the infiltration of various inflammatory cells.
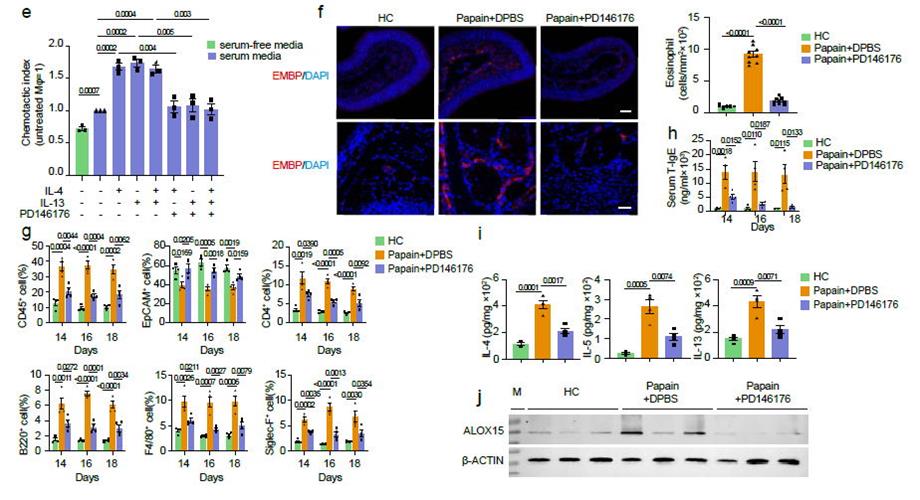
With the scRNA-seq data, the investigators identified that an inhibitor of ALOX15 could reduce the release of proinflammatory chemokines in human macrophages and inhibit the overactivation of type 2 immunity in a mouse model of eosinophilic rhinosinusitis. ALOX15 encodes the enzyme 15-LO, which can oxidize polyunsaturated fatty acids, notably arachidonic acid, and produce a variety of downstream lipid metabolites with proinflammatory activity, which are associated with CRS and asthma. In functional experiments, a 15-LO inhibitor reduced the production on chemokines in IL-4 or IL-13 induced macrophages and the recruitment of CD4+ T cells in vitro. An intragastric 15-LO inhibitor alleviated type 2 inflammation in a mouse model of papain-induced eosinophilic rhinosinusitis in vivo, making it a promising therapeutic target for eCRSwNP (Figure 2).
As their research focused on a complex clinical problem in practical treatment, Prof. Zhang attributed their achievement to a good collaboration of basic research and clinical teams. Their study dissected the heterogeneous immune microenvironment and provides mechanistic insights into the immune pathogenesis of different CRS subtypes. They are quite optimistic for further development and clinical application prospects of products targeting ALOX15 for CRS treatment in future.
This project is supported by National Natural Science Foundation of China, the Natural Science Foundation of Beijing and the CAMS Innovation Fund for Medical Sciences, etc. Prof. Wei He (IBMS, CAMS), Prof. Jianmin Zhang (IBMS, CAMS) and Dr. Wei Lv (PUMCH) are the corresponding authors. Dr. Weiqing Wang (PUMCH), Dr. Yi Xu (IBMS, CAMS) and Dr. Lun Wang (PUMCH) are the co-first authors of this paper.
LINKS: https://www.nature.com/articles/s41590-022-01312-0
In this study, they performed a single-cell level analysis of immune and nonimmune cells in healthy individuals and patients with three subtypes of chronic rhinosinusitis (CRS) and identified some disease-specific cell subsets and molecules that specifically contribute to the pathogenesis. Notably, ALOX15+ macrophages were identified to make a major contribution to the pathogenesis of type 2 immunity in eosinophilic CRS with nasal polyps (eCRSwNP) by secreting chemokines that recruited eosinophils, monocytes and TH2 cells. The findings of this study not only provide a further understanding of the heterogeneous immune microenvironment and the pathogenesis of CRS subtypes but also identified novel targets for the development of therapeutic approaches for the treatment of CRS and potentially other type 2 immunity-mediated diseases.
Human airway chronic inflammatory diseases, including CRS and asthma, share specific mechanisms of pathogenesis and often coexist. Heterogeneous CRS can be divided into different subtypes, including CRS with nasal polyps (CRSwNP) and CRS without nasal polyps (CRSsNP), with the former further divided into eosinophilic CRSwNP (eCRSwNP) and non-eosinophilic CRSwNP (neCRSwNP). Current clinical guidelines still recommend local glucocorticosteroids as primary medication in the management of CRS and asthma, irrespective of disease subtypes. However, because of the heterogeneity of CRS and asthma subtypes with distinct immune responses and inflammatory profiles, such as type 2 and non-type 2 inflammation, the efficacy of these treatments is quite variable. The inflammatory microenvironment in CRS is complex and heterogeneous. Therefore, it would be meaningful to elevate treatment efficacy by modulating clinical therapeutic plan precisely according to systematic understandings for the compositions of immune microenvironment and pathogenesis mechanisms in different CRS subtypes. (Fig1)

Figure 1 Graphic summary of novel insights on the immune microenvironment in eCRSwNP
This work provided a detailed atlas of the immune and nonimmune cell microenvironment in the nasal mucosa of healthy individuals and patients with three heterogeneous subtypes of CRS at single-cell resolution, identifying novel disease-specific cell subsets and clusters, and key molecules associated with distinctive type 2 immunity related to eCRSwNP. The graphic summary illustrates novel insights on different nasal mucosa immune microenvironments between eCRSwNP and healthy controls, and how the type 2 immune response promotes the pathogenesis of nasal polyps in their scRNA-seq study. In eCRSwNP, the hallmarks of pathogenesis included: (1) hyperplasia of basal cells and deficiency of protective cells and molecules, (2) extracellular matrix remodeling dysfunction by fibroblasts, macrophages, vascular endothelial cells, and smooth muscle cells (SMCs), (3) elevated levels of TH2 cells, ILC2s, IL5RA+ plasma cells, and cytotoxic CX3CR1+CD8+ TEFF cells and NK cells, and deficiency of CD8+ TRM cells, (4) defects in the NK-cDC1 immunologic surveillance axis and overactivation of the ALOX15+ cDC2-TH2 axis, and very importantly, (5) enrichment of ALOX15+ macrophages with a prominent chemotactic effect as a driver of the type 2 immunity by promoting the infiltration of various inflammatory cells.

Figure 2 ALOX15 blockade treatment alleviates the type 2 immune response in vitro and in vivo
With the scRNA-seq data, the investigators identified that an inhibitor of ALOX15 could reduce the release of proinflammatory chemokines in human macrophages and inhibit the overactivation of type 2 immunity in a mouse model of eosinophilic rhinosinusitis. ALOX15 encodes the enzyme 15-LO, which can oxidize polyunsaturated fatty acids, notably arachidonic acid, and produce a variety of downstream lipid metabolites with proinflammatory activity, which are associated with CRS and asthma. In functional experiments, a 15-LO inhibitor reduced the production on chemokines in IL-4 or IL-13 induced macrophages and the recruitment of CD4+ T cells in vitro. An intragastric 15-LO inhibitor alleviated type 2 inflammation in a mouse model of papain-induced eosinophilic rhinosinusitis in vivo, making it a promising therapeutic target for eCRSwNP (Figure 2).
As their research focused on a complex clinical problem in practical treatment, Prof. Zhang attributed their achievement to a good collaboration of basic research and clinical teams. Their study dissected the heterogeneous immune microenvironment and provides mechanistic insights into the immune pathogenesis of different CRS subtypes. They are quite optimistic for further development and clinical application prospects of products targeting ALOX15 for CRS treatment in future.
This project is supported by National Natural Science Foundation of China, the Natural Science Foundation of Beijing and the CAMS Innovation Fund for Medical Sciences, etc. Prof. Wei He (IBMS, CAMS), Prof. Jianmin Zhang (IBMS, CAMS) and Dr. Wei Lv (PUMCH) are the corresponding authors. Dr. Weiqing Wang (PUMCH), Dr. Yi Xu (IBMS, CAMS) and Dr. Lun Wang (PUMCH) are the co-first authors of this paper.
LINKS: https://www.nature.com/articles/s41590-022-01312-0


