Dr. Feng Shao’s Team Establishes a New Conceptual Framework for Cancer Immunotherapy
Source:Jiaqi Liu
2026-03-05
On December 10, 2025, Dr. Feng Shao’s team at the National Institute of Biological Sciences, Beijing (NIBS)/ Tsinghua Institute of Multidisciplinary Biomedical Research (TIMBR), Tsinghua University, published a research article in Nature entitled “Agonists for cytosolic bacterial receptor ALPK1 induce antitumour immunity.” This study is the first to systematically demonstrate that activation of the cytosolic innate immune receptor ALPK1 elicits robust antitumor immune responses, establishing a new conceptual framework for cancer immunotherapy.
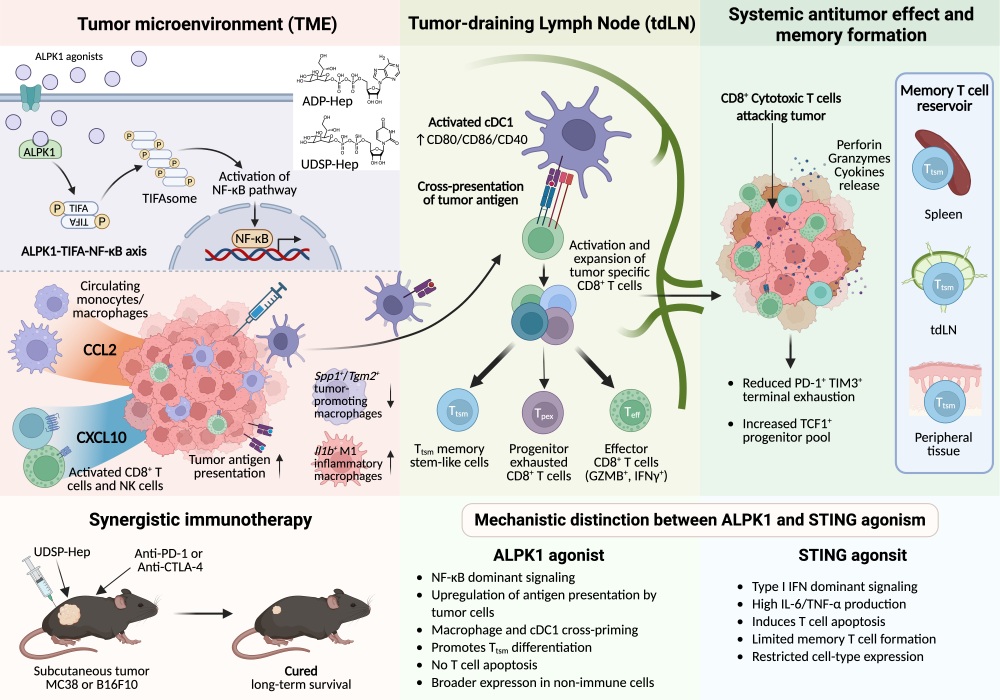
This work builds upon the Shao laboratory’s previous discovery that ALPK1 recognizes the bacterial metabolite ADP-heptose (ADP-Hep). Extending this mechanistic insight to tumor immunity, the team investigated the therapeutic potential of ALPK1 activation in multiple mouse tumor models. Intratumoral administration of the natural ligand ADP-Hep significantly suppressed tumor growth in an ALPK1-dependent manner, providing initial proof-of-concept for the antitumor activity of ALPK1 agonists. However, ADP-Hep exhibits poor pharmacological stability, limiting its translational potential. To overcome this challenge, the researchers employed medicinal chemistry optimization to develop a novel synthetic agonist, UDSP-Hep. Structural modification markedly improved its stability and enhanced its potency approximately 50-fold relative to the natural ligand, establishing UDSP-Hep as a powerful tool compound for efficacy and mechanistic studies.
Therapeutically, UDSP-Hep demonstrated striking antitumor efficacy. It inhibited or eradicated tumors across multiple models and induced durable protective immune memory lasting up to one year in cured mice, effectively preventing tumor rechallenge. Importantly, combination therapy with anti-CTLA-4 or anti-PD-1 antibodies produced strong synergistic effects. This combination not only significantly prolonged survival but also converted immune checkpoint inhibitor–resistant “cold tumors” into responsive tumors, suggesting a promising strategy to overcome resistance in clinical immunotherapy.
Mechanistic investigations revealed that the antitumor effects of UDSP-Hep depend on CD8+ T cells, dendritic cells, and macrophages, and involve the chemokines CXCL10 and CCL2. UDSP-Hep directly enhanced dendritic cell activation and antigen cross-presentation, thereby promoting efficient priming of tumor-specific CD8+ T cells. Single-cell transcriptomic analysis further showed that UDSP-Hep remodels the tumor immune microenvironment by increasing the proportion of progenitor-like exhausted T cells, which possess superior proliferative and persistence capacity, while reducing terminally exhausted T cells. This cellular reprogramming provides a mechanistic basis for sustained antitumor immunity.
Compared with extensively studied STING or TLR7/8 agonists, ALPK1 agonists exhibit several distinguishing advantages. ALPK1 displays broader expression, enabling direct enhancement of antigen presentation in tumor cells themselves. Moreover, ALPK1 activation induces lower levels of potentially deleterious pro-inflammatory cytokines such as IL-6 and TNF-α. Importantly, it promotes a higher proportion of tumor-specific memory T cells without triggering apoptosis of activated T cells. Collectively, these features suggest an improved safety profile and enhanced durability of immune protection.
In summary, this study identifies ALPK1 as a highly promising therapeutic target for cancer immunotherapy, reports the development of a potent and stable agonist (UDSP-Hep), and systematically elucidates its mechanisms and unique immunological advantages. These findings provide a novel strategy to address current limitations in immunotherapy and lay a strong theoretical foundation for future clinical translation.
Dr. Xiaoying Tian and Ph.D. candidate Jiaqi Liu from Dr. Feng Shao’s laboratory at NIBS are co–first authors of the study. Additional contributors include Dr. Yuxi Li from the Li Chao laboratory at NIBS; Dr. Yupeng Wang, Ph.D. candidate Yuanyuan Hanyu Luo, Dr. Huabin He, and Dr. Yang She from the Feng Shao laboratory at NIBS; Dr. Yan Ma from the Metabolomics Center at NIBS; Dr. Jingjin Ding from the Institute of Biophysics, Chinese Academy of Sciences; Dr. Ping Zhou from Sun Yat-sen University Cancer Center; and Dr. Chao Li from NIBS. Dr. Feng Shao is the corresponding author. This work was supported by the Basic Science Center Project of the National Natural Science Foundation of China (NSFC), the Strategic Priority Research Program of Chinese Academy of Sciences, the National Key Research and Development Program of China and the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences. Dr. Feng Shao is also supported by the Tencent New Cornerstone Investigator Program.
Article link: https://www.nature.com/articles/s41586-025-09828-9
This work builds upon the Shao laboratory’s previous discovery that ALPK1 recognizes the bacterial metabolite ADP-heptose (ADP-Hep). Extending this mechanistic insight to tumor immunity, the team investigated the therapeutic potential of ALPK1 activation in multiple mouse tumor models. Intratumoral administration of the natural ligand ADP-Hep significantly suppressed tumor growth in an ALPK1-dependent manner, providing initial proof-of-concept for the antitumor activity of ALPK1 agonists. However, ADP-Hep exhibits poor pharmacological stability, limiting its translational potential. To overcome this challenge, the researchers employed medicinal chemistry optimization to develop a novel synthetic agonist, UDSP-Hep. Structural modification markedly improved its stability and enhanced its potency approximately 50-fold relative to the natural ligand, establishing UDSP-Hep as a powerful tool compound for efficacy and mechanistic studies.
Therapeutically, UDSP-Hep demonstrated striking antitumor efficacy. It inhibited or eradicated tumors across multiple models and induced durable protective immune memory lasting up to one year in cured mice, effectively preventing tumor rechallenge. Importantly, combination therapy with anti-CTLA-4 or anti-PD-1 antibodies produced strong synergistic effects. This combination not only significantly prolonged survival but also converted immune checkpoint inhibitor–resistant “cold tumors” into responsive tumors, suggesting a promising strategy to overcome resistance in clinical immunotherapy.
Mechanistic investigations revealed that the antitumor effects of UDSP-Hep depend on CD8+ T cells, dendritic cells, and macrophages, and involve the chemokines CXCL10 and CCL2. UDSP-Hep directly enhanced dendritic cell activation and antigen cross-presentation, thereby promoting efficient priming of tumor-specific CD8+ T cells. Single-cell transcriptomic analysis further showed that UDSP-Hep remodels the tumor immune microenvironment by increasing the proportion of progenitor-like exhausted T cells, which possess superior proliferative and persistence capacity, while reducing terminally exhausted T cells. This cellular reprogramming provides a mechanistic basis for sustained antitumor immunity.
Compared with extensively studied STING or TLR7/8 agonists, ALPK1 agonists exhibit several distinguishing advantages. ALPK1 displays broader expression, enabling direct enhancement of antigen presentation in tumor cells themselves. Moreover, ALPK1 activation induces lower levels of potentially deleterious pro-inflammatory cytokines such as IL-6 and TNF-α. Importantly, it promotes a higher proportion of tumor-specific memory T cells without triggering apoptosis of activated T cells. Collectively, these features suggest an improved safety profile and enhanced durability of immune protection.

In summary, this study identifies ALPK1 as a highly promising therapeutic target for cancer immunotherapy, reports the development of a potent and stable agonist (UDSP-Hep), and systematically elucidates its mechanisms and unique immunological advantages. These findings provide a novel strategy to address current limitations in immunotherapy and lay a strong theoretical foundation for future clinical translation.
Dr. Xiaoying Tian and Ph.D. candidate Jiaqi Liu from Dr. Feng Shao’s laboratory at NIBS are co–first authors of the study. Additional contributors include Dr. Yuxi Li from the Li Chao laboratory at NIBS; Dr. Yupeng Wang, Ph.D. candidate Yuanyuan Hanyu Luo, Dr. Huabin He, and Dr. Yang She from the Feng Shao laboratory at NIBS; Dr. Yan Ma from the Metabolomics Center at NIBS; Dr. Jingjin Ding from the Institute of Biophysics, Chinese Academy of Sciences; Dr. Ping Zhou from Sun Yat-sen University Cancer Center; and Dr. Chao Li from NIBS. Dr. Feng Shao is the corresponding author. This work was supported by the Basic Science Center Project of the National Natural Science Foundation of China (NSFC), the Strategic Priority Research Program of Chinese Academy of Sciences, the National Key Research and Development Program of China and the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences. Dr. Feng Shao is also supported by the Tencent New Cornerstone Investigator Program.
Article link: https://www.nature.com/articles/s41586-025-09828-9


