Chinese Researchers Construct the Human Immune Aging Clock and Identify a Decelerator of T Cell Senescence
Source:Weiqi Zhang
2026-04-20
On April 14, 2026, a collaborative study led by Weiqi Zhang (China National Center for Bioinformation), Guang-Hui Liu (Institute of Zoology, Chinese Academy of Sciences), Feng Zhang (Quzhou Affiliated Hospital of Wenzhou Medical University), Lan Jiang (China National Center for Bioinformation), Yun-Gui Yang (China National Center for Bioinformation), and Jing Qu (Institute of Zoology, Chinese Academy of Sciences) was published online in Immunity, entitled “Human immune aging clock identifies RUNX1 as a decelerator of T cell senescence”. In this work, the authors constructed a high-precision Human Immune Aging Clock (HIAC), systematically elucidated the multi-scale dynamics of immunosenescence, and identified the transcription factor RUNX1 as a functional “brake” on T cell senescence. Using primary human immune cells and humanized mouse models, they further demonstrated that RUNX1 overexpression can effectively rescue T cell senescence. This study establishes a comprehensive evidence chain from quantitative assessment to causal intervention, advancing immunosenescence research into a quantifiable, modelable, and targetable paradigm.
Traditional approaches to immunosenescence have largely relied on single biomarkers or bulk transcriptomic analyses, which fail to capture subtype-specific alterations of immune cells. To address this limitation, the research team profiled peripheral blood mononuclear cells (PBMCs) from 230 healthy individuals spanning a 60-year age range, generating a high-resolution single-cell atlas of approximately 1.2 million cells and identifying 24 immune cell subtypes. The analysis revealed that aging induces a profound remodeling of the immune landscape, characterized by a marked attrition of the naïve T cell pool and an expansion of exhausted T cells and monocytes, reflecting a state in which immune exhaustion coexists with chronic inflammation.
Building on these data, the authors established a multi-layered immune aging clock framework, including pAge based on cell proportions, tAge based on cell type-specific transcriptomes, TCRAge based on T cell receptor repertoire features, and an integrated multi-modal clock (immAge). Among these, immAge achieved a Mean Absolute Error (MAE) of 5.66 years in predicting chronological age, while T cell-based clock models exhibited the best predictive performance across all cell types. These findings demonstrate at single-cell resolution that T cells are the most sensitive indicators of peripheral immunosenescence.
The study further extended immune aging assessment from population-level patterns to individualized characterization by introducing the concept of “immune aging pace”, defined as the residual of predicted immune age from chronological age based on linear regression. This metric enabled the stratification of individuals into “age accelerators” and “age decelerators”. Individuals with decelerated immune aging exhibited a more youthful immune profile, including higher proportions of naïve T cells, diminished senescence- and inflammation-associated transcriptional signatures, and plasma metabolomic profiles enriched for immunomodulatory and antioxidant metabolites. These individuals also exhibited favorable physiological homeostasis, including optimized glycemic control, better liver function, and enhanced cardiopulmonary capacity. These results establish a direct link between immune aging dynamics and systemic health, suggesting that immune age may serve as a more sensitive predictor of functional decline than chronological age.
Through gene regulatory network analysis, the authors found that the downstream targets of the transcription factor RUNX1 carried the highest weights in the immune aging clock. Further investigations revealed that RUNX1 functions as a youth-associated factor in T cells, where maintaining its expression helps counteract T cell senescence, whereas its overexpression can reverse aging-associated phenotypes. In both CD4⁺ and CD8⁺ T cells, RUNX1 protein levels decline with age. Functional experiments confirmed that RUNX1 is a key regulator of T cell senescence: deletion of RUNX1 in young donor T cells induced cell cycle arrest, activation of the senescence-associated secretory phenotype (SASP), telomere attrition in CD8⁺ T cells, and reduced cytotoxic capacity; conversely, overexpression of RUNX1 in aged T cells reversed these senescence-associated features, including restoration of the co-stimulatory molecule CD27 (thereby enhancing T cell activation and memory potential) and elongation of telomeres. Mechanistically, RUNX1 binds to and activates a network of key genes involved in T cell activation and immune responses in CD8⁺ T cells, thereby maintaining a youthful transcriptional program. In vivo adoptive transfer experiments further validated these findings, as aged CD8⁺ T cells overexpressing RUNX1 maintained lower levels of senescence markers and higher CD27 expression within immunodeficient mice, demonstrating that RUNX1 confers protection against immunosenescence in a physiological context.
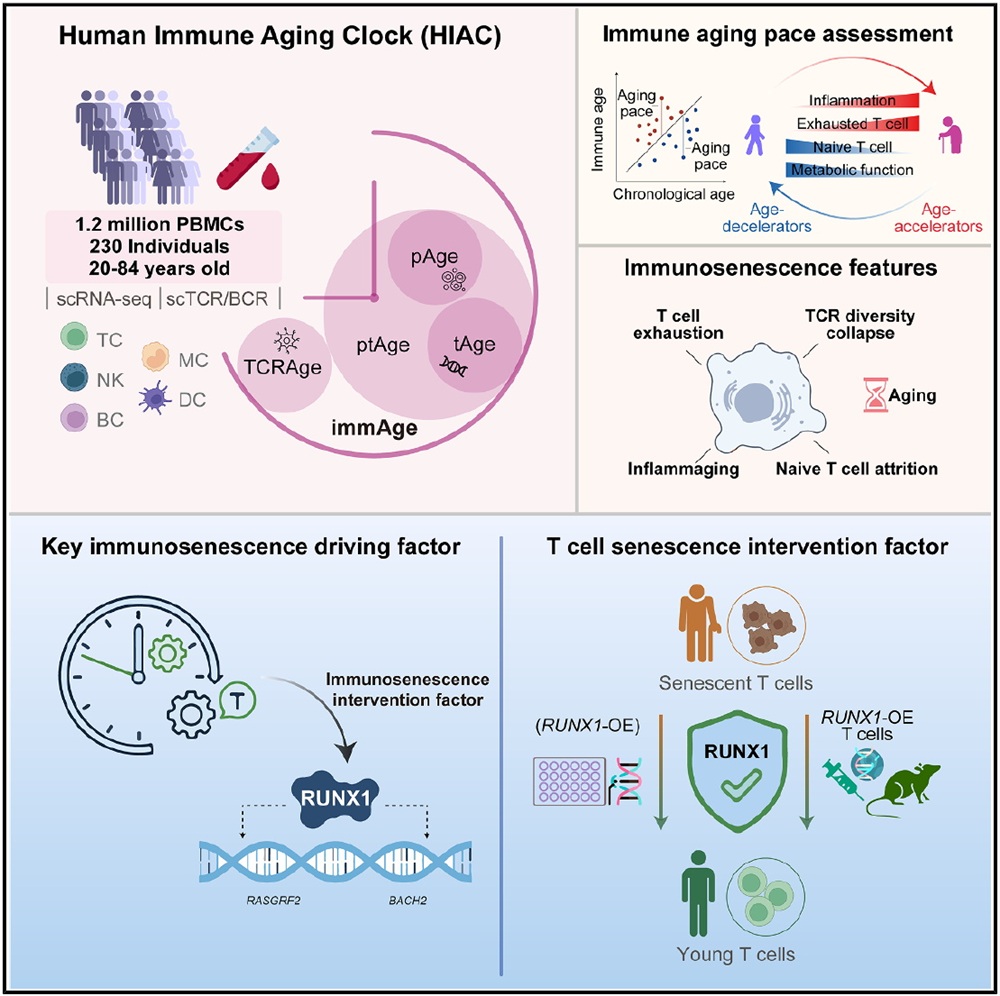
Based on single-cell resolution, this study establishes a comprehensive human immune aging clock that quantitatively captures immunosenescence across multiple dimensions, including cellular composition, transcriptional regulation, and immune repertoire dynamics, and identifies T cells as the core drivers of immune aging. By further identifying RUNX1 as a targetable regulator, the study proposes transcription factor reactivation as a novel strategy for delaying immunosenescence. These findings transform immunosenescence into a quantifiable and intervenable systems-level framework, providing a theoretical foundation for developing interventions to mitigate age-related immune decline, particularly in elderly populations. Notably, this work offers a potential avenue to enhance the durability and efficacy of T cell–based immunotherapies (e.g., CAR-T) in elderly populations. Future studies are needed to further elucidate the context-specific regulatory roles of RUNX1 across diverse immune cell types and aging-associated diseases, as well as its interactions with other aging-related pathways. As a youth-associated factor in human T cells, RUNX1 warrants further investigation into its potential to enhance T cell function and to facilitate the clinical translation of immune rejuvenation strategies, ultimately contributing to the development of immunological solutions for healthy aging.
Weiqi Zhang (China National Center for Bioinformation), Guang-Hui Liu (Institute of Zoology, Chinese Academy of Sciences), Feng Zhang (Quzhou Affiliated Hospital of Wenzhou Medical University), Lan Jiang and Yun-Gui Yang (China National Center for Bioinformation), and Jing Qu (Institute of Zoology, Chinese Academy of Sciences) are the corresponding authors of this study. Jiale Ping (China National Center for Bioinformation), Qin Qiao (China National Center for Bioinformation), Dan-Dan Gao (Quzhou Affiliated Hospital of Wenzhou Medical University), Yun Li (Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences), Yanling Fan (China National Center for Bioinformation), Yutong Tian (China National Center for Bioinformation), Muzhao Xiong (China National Center for Bioinformation), Quan Zheng and Beier Jiang (Quzhou Affiliated Hospital of Wenzhou Medical University) are co-first authors of this study.
Article Link: https://doi.org/10.1016/j.immuni.2026.02.007
Traditional approaches to immunosenescence have largely relied on single biomarkers or bulk transcriptomic analyses, which fail to capture subtype-specific alterations of immune cells. To address this limitation, the research team profiled peripheral blood mononuclear cells (PBMCs) from 230 healthy individuals spanning a 60-year age range, generating a high-resolution single-cell atlas of approximately 1.2 million cells and identifying 24 immune cell subtypes. The analysis revealed that aging induces a profound remodeling of the immune landscape, characterized by a marked attrition of the naïve T cell pool and an expansion of exhausted T cells and monocytes, reflecting a state in which immune exhaustion coexists with chronic inflammation.
Building on these data, the authors established a multi-layered immune aging clock framework, including pAge based on cell proportions, tAge based on cell type-specific transcriptomes, TCRAge based on T cell receptor repertoire features, and an integrated multi-modal clock (immAge). Among these, immAge achieved a Mean Absolute Error (MAE) of 5.66 years in predicting chronological age, while T cell-based clock models exhibited the best predictive performance across all cell types. These findings demonstrate at single-cell resolution that T cells are the most sensitive indicators of peripheral immunosenescence.
The study further extended immune aging assessment from population-level patterns to individualized characterization by introducing the concept of “immune aging pace”, defined as the residual of predicted immune age from chronological age based on linear regression. This metric enabled the stratification of individuals into “age accelerators” and “age decelerators”. Individuals with decelerated immune aging exhibited a more youthful immune profile, including higher proportions of naïve T cells, diminished senescence- and inflammation-associated transcriptional signatures, and plasma metabolomic profiles enriched for immunomodulatory and antioxidant metabolites. These individuals also exhibited favorable physiological homeostasis, including optimized glycemic control, better liver function, and enhanced cardiopulmonary capacity. These results establish a direct link between immune aging dynamics and systemic health, suggesting that immune age may serve as a more sensitive predictor of functional decline than chronological age.
Through gene regulatory network analysis, the authors found that the downstream targets of the transcription factor RUNX1 carried the highest weights in the immune aging clock. Further investigations revealed that RUNX1 functions as a youth-associated factor in T cells, where maintaining its expression helps counteract T cell senescence, whereas its overexpression can reverse aging-associated phenotypes. In both CD4⁺ and CD8⁺ T cells, RUNX1 protein levels decline with age. Functional experiments confirmed that RUNX1 is a key regulator of T cell senescence: deletion of RUNX1 in young donor T cells induced cell cycle arrest, activation of the senescence-associated secretory phenotype (SASP), telomere attrition in CD8⁺ T cells, and reduced cytotoxic capacity; conversely, overexpression of RUNX1 in aged T cells reversed these senescence-associated features, including restoration of the co-stimulatory molecule CD27 (thereby enhancing T cell activation and memory potential) and elongation of telomeres. Mechanistically, RUNX1 binds to and activates a network of key genes involved in T cell activation and immune responses in CD8⁺ T cells, thereby maintaining a youthful transcriptional program. In vivo adoptive transfer experiments further validated these findings, as aged CD8⁺ T cells overexpressing RUNX1 maintained lower levels of senescence markers and higher CD27 expression within immunodeficient mice, demonstrating that RUNX1 confers protection against immunosenescence in a physiological context.

Based on single-cell resolution, this study establishes a comprehensive human immune aging clock that quantitatively captures immunosenescence across multiple dimensions, including cellular composition, transcriptional regulation, and immune repertoire dynamics, and identifies T cells as the core drivers of immune aging. By further identifying RUNX1 as a targetable regulator, the study proposes transcription factor reactivation as a novel strategy for delaying immunosenescence. These findings transform immunosenescence into a quantifiable and intervenable systems-level framework, providing a theoretical foundation for developing interventions to mitigate age-related immune decline, particularly in elderly populations. Notably, this work offers a potential avenue to enhance the durability and efficacy of T cell–based immunotherapies (e.g., CAR-T) in elderly populations. Future studies are needed to further elucidate the context-specific regulatory roles of RUNX1 across diverse immune cell types and aging-associated diseases, as well as its interactions with other aging-related pathways. As a youth-associated factor in human T cells, RUNX1 warrants further investigation into its potential to enhance T cell function and to facilitate the clinical translation of immune rejuvenation strategies, ultimately contributing to the development of immunological solutions for healthy aging.
Weiqi Zhang (China National Center for Bioinformation), Guang-Hui Liu (Institute of Zoology, Chinese Academy of Sciences), Feng Zhang (Quzhou Affiliated Hospital of Wenzhou Medical University), Lan Jiang and Yun-Gui Yang (China National Center for Bioinformation), and Jing Qu (Institute of Zoology, Chinese Academy of Sciences) are the corresponding authors of this study. Jiale Ping (China National Center for Bioinformation), Qin Qiao (China National Center for Bioinformation), Dan-Dan Gao (Quzhou Affiliated Hospital of Wenzhou Medical University), Yun Li (Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences), Yanling Fan (China National Center for Bioinformation), Yutong Tian (China National Center for Bioinformation), Muzhao Xiong (China National Center for Bioinformation), Quan Zheng and Beier Jiang (Quzhou Affiliated Hospital of Wenzhou Medical University) are co-first authors of this study.
Article Link: https://doi.org/10.1016/j.immuni.2026.02.007


